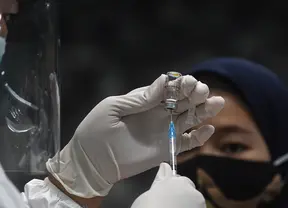
Indonesia has approved China's Sinovac Biotech's COVID-19 vaccine for its elderly population, reported Reuters citing the country's food and drugs authority (BPOM).According to Reuters, the BPOM gran
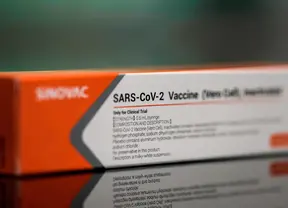
Sinovac Biotech received a conditional commercial approval from the National Medical Products Administration for its COVID-19 vaccine in China Friday, according to the

The World Health Organization (WHO) said on Tuesday that it's evaluating data submitted by China's Sinovac and Sinopharm on their COVID-19 vaccines for emergency use.

02:39 Clinical trials in Brazil are shedding new light on China's Sinovac COVID-19 vaccine.The efficacy rate dropped to just over 50 percent in late analysis of trials.Sao Paulo state

China's COVID-19 vaccine developed by Sinovac Biotech may become a new candidate and be subjected to evaluation by the European Union, Nicola Magrini, general director of the Italian Medicines Agency

Indonesian President Joko Widodo on Wednesday received the COVID-19 vaccine shot developed by Chinese biopharmaceutical company Sinovac Biotech.

Indonesian President Joko Widodo on Wednesday received the COVID-19 vaccine shot developed by Chinese biopharmaceutical company Sinovac Biotech.

A World Health Organization (WHO) team is in China currently working with producers of the Sinovac and Sinopharm vaccines to assess compliance with international quality manufacturing practices ahead
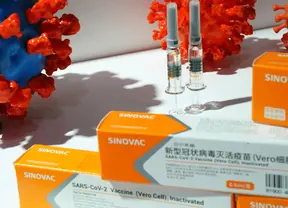
Indonesia's Food and Drug Authority on Monday green-lighted the emergency use of the COVID-19 vaccine produced by Chinese biopharmaceutical company Sinovac Biotech, with vaccinations of high-risk gro