02:52
**Editor's note: **This is the weekly wrap-up of Chinese COVID-19 vaccines' contribution to the global fight against the virus for May 3 - May 9, 2021.
The World Health Organization (WHO) announced the approval of the Sinopharm vaccine for Emergency Use Listing (EUL) on Friday, making it the sixth vaccine to receive WHO validation for safety, efficacy and quality.
The approval reflects the international community's recognition of the research and development, production and safety of the Sinopharm vaccines in line with international standards.Most importantly, after the WHO's approval, the vaccines will be able to contribute more to the global fight against the pandemic as they will enter the global procurement and supply system.
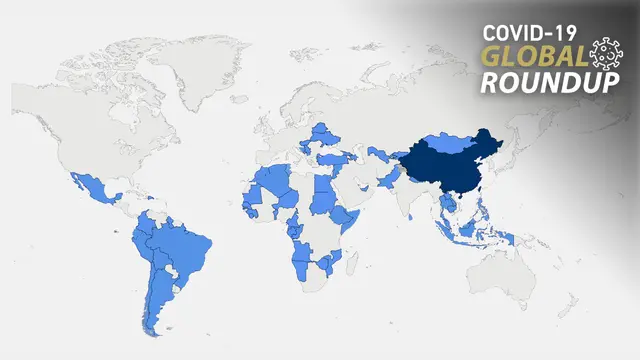
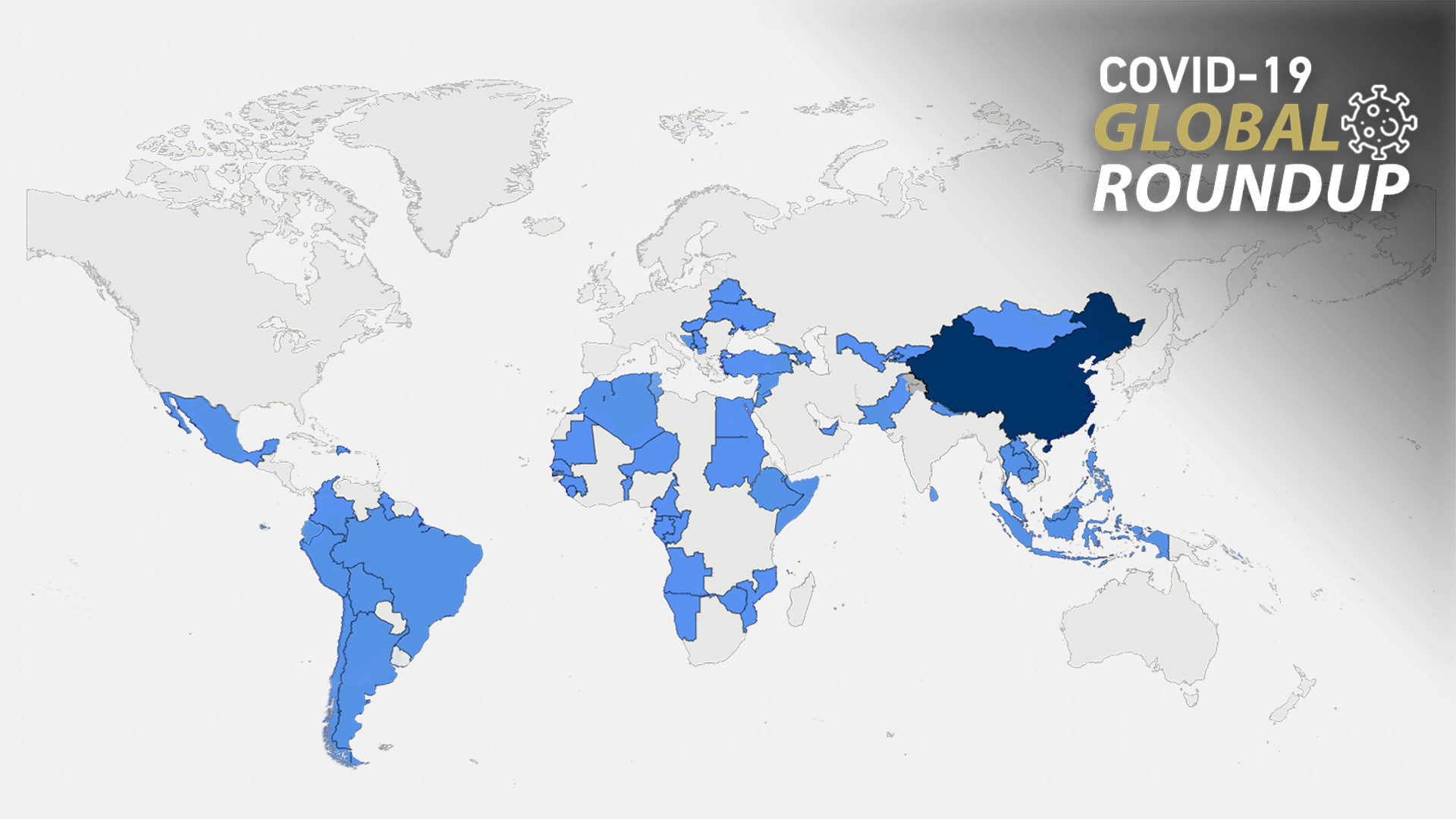
So far, China has providesvaccines to more than 100 countries and international organizations amid a resurgence in COVID-19 outbreaks around the world.

Countries including Pakistan, Azerbaijan, the Philippines, North Macedoniaand Hungary are among the latest to receive vaccines from China. Azerbaijan's acting health minister Teymur Musayev on Thursday hailed the CoronaVac vaccine made by China's Sinovac Biotech as "completely safe." He said that 1.6 million people in the country have got their first dose of the Chinese-made vaccine so far, while 600,000 people have received both.
As the first country in the EU to use the Sinopharm vaccine, Hungary, in turn, has achieved one of the EU's highest vaccination rates. Hungary announced on April 29that it had received over 2 million dosesof vaccine shipments as part of a contract for 5 million doses from China.
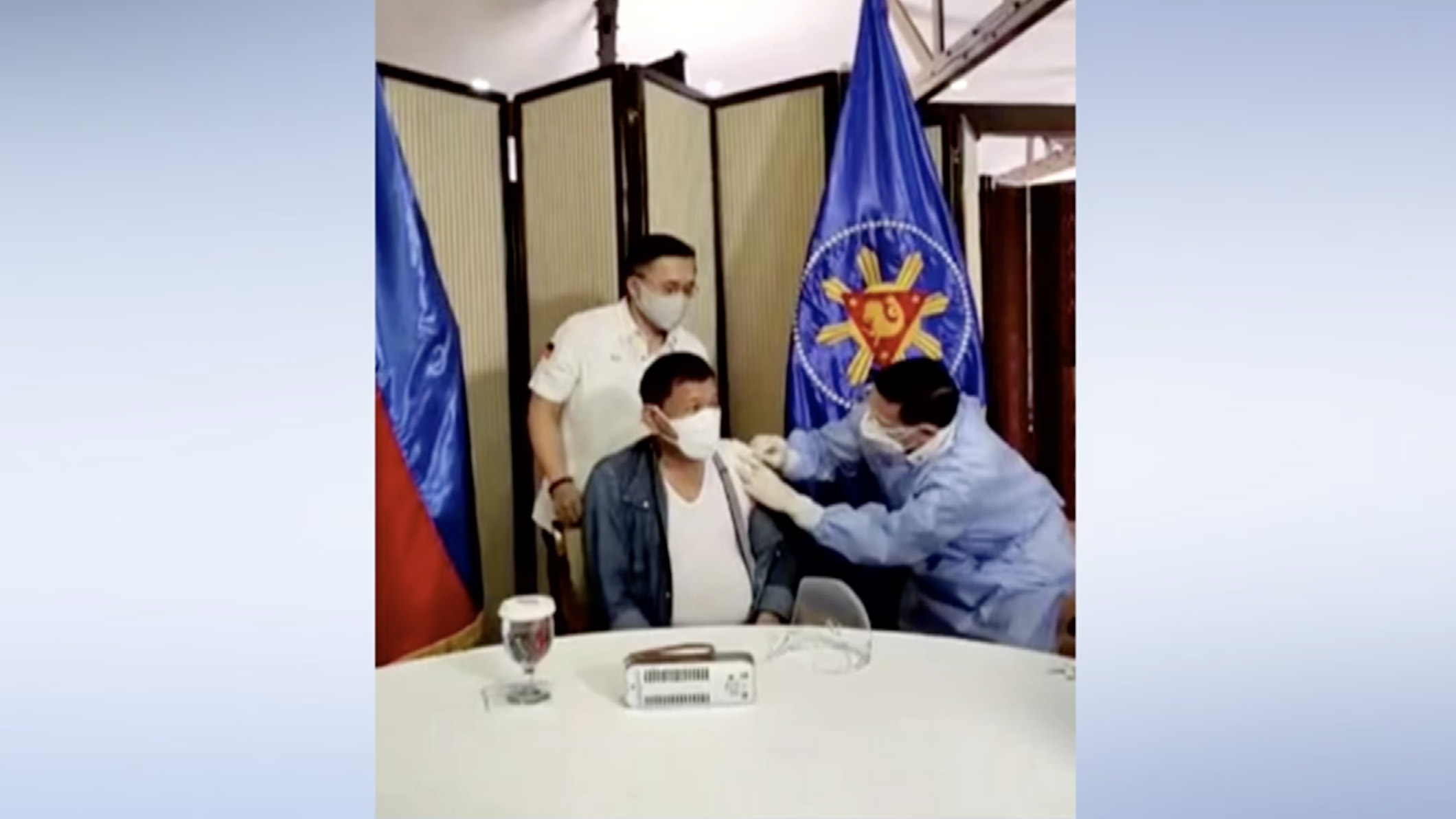
Meanwhile, several world leaders have also taken Chinese vaccines during the past week, such as the Dominican Republic's president Luis Abinader, Philippine PresidentRodrigo Duterte and Prime Minister of North Macedonia Zoran Zaev.
"I feel good, and I have been expecting this shot, vaccination a long time ago," Duterte, 76, said on May 3 before receiving the Sinopharm vaccine and encouraged Filipinos to get vaccinated.
00:24
Zaev made an equally positive assessment after being vaccinated, saying that "I feel great and will continue my daily work agenda." He also encouraged all citizens to register for the vaccination, adding that the vaccines are safe, checked and tested.
Besides, Chinese vaccine makers are cooperating with more countries. Chinese pharmaceutical giant Sinopharm will jointly produce its COVID-19 vaccine jabs with Argentine companies, the Chinese embassy in Argentina said on May 3. Chinese Ambassador to Argentina Zou Xiaoli said the two sides are discussing technical details of producing the vaccines and will start the process as soon as possible.
The first batch of China's CanSino COVID-19 vaccines that the Pakistani government purchased in bulk arrived in Pakistan on Tuesday. The Special Assistant to the Pakistani Prime Minister on Health Faisal Sultan said on May 3 that the local production of the single-dose CanSino COVID-19 vaccine is about to commence in the country to facilitate its vaccination drive.
China has also pledged to provide 10 million doses of its COVID-19 vaccines to the global vaccine distribution initiative known as the COVID-19 Vaccines Global Access (COVAX).
 简体中文
简体中文